Experiment # 2 DISSOLVED OXYGEN
TITLE
To determine the amount of Dissolved Oxygen (DO) in water.
DISSOLVED OXYGEN
The dissolved oxygen (DO) is oxygen that is dissolved in water. The oxygen dissolves by diffusion from the surrounding air; aeration of water that has tumbled over falls and rapids; and as a waste product of photosynthesis. A simplified formula is given below:
Photosynthesis (in the presence of light and chlorophyll):
|
Carbon dioxide |
+ |
Water |
————–> |
Oxygen |
+ |
Carbon-rich foods |
|
CO2 |
H2O |
O2 |
C6H12O6 |
Fish and aquatic animals cannot split oxygen from water (H2O) or other oxygen-containing compounds. Only green plants and some bacteria can do that through photosynthesis and similar processes. Virtually all the oxygen we breathe is manufactured by green plants. A total of three-fourths of the earth’s oxygen supply is produced by phytoplankton in the oceans.
- Fish, invertebrates, plants, and aerobic bacteria all require oxygen for respiration.
- Much of the dissolved oxygen in water comes from the atmosphere. After dissolving at the surface, oxygen is distributed by current and turbulence. Algae and rooted aquatic plants also deliver oxygen to water through photosynthesis.
- The main factor contributing to changes in dissolved oxygen levels is the build-up of organic wastes. Decay of organic wastes consumes oxygen and is often concentrated in summer, when aquatic animals require more oxygen to support higher metabolisms.
- Depletions in dissolved oxygen can cause major shifts in the kinds of aquatic organisms found in water bodies.
- Temperature, pressure, and salinity affect the dissolved oxygen capacity of water. The ratio of the dissolved oxygen content (ppm) to the potential capacity (ppm) gives the percent saturation, which is an indicator of water quality.
METHODS OF DETERMINATION OF DO IN WATER:
(1) Winkler Method
(2) Azide modification of Winkler Method
(3) Rideal Stewart method
(4) DO-meters
(1) Winkler Method
Its principle is that oxygen oxidizes manganese (Mn+2) to higher oxidation state then this higher oxidation state manganese converts iodide ion (I-1) to iodine I2 and amount of free iodide librated is equivalent to DO.
| Mn+2 + 2OH-1 | Mn(OH)2 | |
| Mn(OH)2 + 1/2O2 | MnO2 + H2O | |
| MnO2 + 2I-1 + 4H+1 | Mn+2 + I2 + H2O | |
| Then by titration with S2O3-2, thiosulphate ion (thiosulphate ion comes from sodium | ||
| thiosulphate), iodine can be calculated. | ||
| 2S2O3-1 + I2 | S4O6-2 + 2I-1 | |
| If we use 0.025 N Na2S2O3 then 1ml of titrant = 1mg of DO | ||
If nitrites NO2-1 will present in water, they will change the results because they convert iodide ions to iodine before performance of experiment. To remove NO2-1 sodium azides are added.
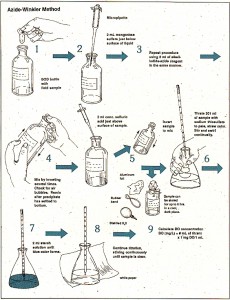
PROCEDURE
- Take BOD bottle (300 ml volume)
- Fill the bottle with water sample.
- Add 1 ml MnSO4 solution to it and mix uniformly with the help of pipette.
- Add 1ml alkali azide iodide solution. On addition, if white ppts. are formed, then there is no DO in water. Formation of Reddish brown ppts. Indicates the presence of DO.
- If Reddish brown ppts are formed, stopper the bottle and shake it upside down for 20 times and allow the ppts. To settle down for about 2 inches.
- Add 1 ml Concentrated H2SO4 and again shake for about 8 times.
- Take 200 ml of this water sample in a titration flask and titrate it with 0.025 N Na2S2O3 till the appearance of light yellow color.
- Then add 1ml starch solution. The color of solution becomes blue on this addition.
- Again titrate it with 0.025N Na2S2O3 till the disappearance of blue color.
- Note the volume of titrant used. ml of titrant used = DO in mg/liter
The above formula is applicable if we use 200ml of water sample solution and 0.025N Na2S2O3. General formula is given by
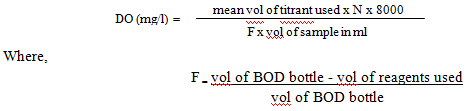
OBSERVATIONS AND CALCULATIONS
|
Sample # |
Sample Description |
Sample Volume (ml) |
Volume of Titrant Na2S2O3 used (ml) |
Dissolved Oxygen (mg/liter) |
Mean Dissolved Oxygen (mg/liter) |
|
1 |
Lab Prepared |
100 |
2.6 |
7.6 |
5.118 |
|
200 |
5 |
||||
|
2 |
Lab Prepared |
100 |
3.1 |
9.7 |
6.532 |
|
200 |
6.6 |
||||
|
3 |
Lab Prepared |
300 |
No Oxygen |
||
Comments:
Polluted water contains less oxygen than fresh water because polluted water is more saturated as compared to fresh water. In sample 3, after the addition on 1ml of MnSO4 White precipitates are formed, which shows that there is no oxygen present in the sample. So we discard the sample and it shows that the sample is highly polluted.
Questions:
1) What are the factors upon which solubility of oxygen depends?
Temperature inversely controls the solubility of oxygen in water; as temperature increases, oxygen is less soluble.
pressure increases due to weather or elevation changes, oxygen solubility increases.
Salinity reduces the solubility of oxygen in water.
aquatic vegetation, microorganisms, and algae consume oxygen at all hours of the day and night.
 Water solubility of oxygen at 25oC and pressure = 1 bar is at 40 mg/L water. In air with a normal composition the oxygen partial pressure is 0.2 atm. This results in dissolution of 40 . 0.2 = 8 mg O2/L in water that comes in contact with air. Oxygen solubility is strongly temperature dependent and decreases at higher temperatures. Oxygen solubility is negatively correlated with the amount of dissolved solids. Consequently, oxygen solubility in freshwater exceeds that in seawater by 1-3 mg/L, depending on temperature. The saturation constant in rivers and lakes in mountainous areas is usually lower than in lowlands, because it is pressure dependent
Water solubility of oxygen at 25oC and pressure = 1 bar is at 40 mg/L water. In air with a normal composition the oxygen partial pressure is 0.2 atm. This results in dissolution of 40 . 0.2 = 8 mg O2/L in water that comes in contact with air. Oxygen solubility is strongly temperature dependent and decreases at higher temperatures. Oxygen solubility is negatively correlated with the amount of dissolved solids. Consequently, oxygen solubility in freshwater exceeds that in seawater by 1-3 mg/L, depending on temperature. The saturation constant in rivers and lakes in mountainous areas is usually lower than in lowlands, because it is pressure dependent
2) Why starch is added when light yellow colour appears?
The titration is actually titrating iodine, from yellow to clear. Since the yellow to clear change is very hard to see we add starch, which turns blue in the presence of iodine. Once all the iodine has been titrated out the starch goes clear. The blue to clear is much easier to see than yellow to clear. The reason that we titrate some of the iodine out (titrate to faint yellow) before adding starch is two-fold:
- The starch stays dark blue right up until it goes clear, unlike most titrations where the color gradually moves toward the endpoint. Therefore, it is easy to become complacent during the titration and add an excess amount of titrant (overshooting the endpoint), thinking that you are far from the endpoint because the color is not changing.
- Also starch can be partially decomposed by a large amount of iodine. Therefore, the starch should not be added until the bulk of the iodine has been reduced (titrated out).
So, for both of these reasons, the sample should be titrated to a faint yellow (the exact shade does not matter) before adding the starch
3) Write the significance of this test in Environmental Engineering.
Dissolved oxygen (DO) levels in natural and wastewaters depend on the physical, chemical, and biochemical activities in the water body. The analysis for DO is a key test in water pollution and waste treatment process control.
- Oxygen is critical to the survival of aquatic plants and animals, and a shortage of dissolved oxygen is not only a sign of pollution, it is harmful to the fish. Some aquatic species are more sensitive to oxygen depletion than others, but some general guidelines to consider when analyzing test results are:
|
5-6 ppm |
Sufficient for most species |
|
< 3 ppm |
Stressful to most aquatic species |
|
< 2 ppm |
Fatal to most species |
Because of its importance to the fish’s survival, aquaculturists, or “fish farmers,” and aquarists use the dissolved oxygen test as a primary indicator of their system’s ability to support healthy fish.
Dissolved oxygen analysis can be used to determine:
- the health or cleanliness of a lake or stream,
- the amount and type of biomass a freshwater system can support,
- the amount of decomposition occurring in the lake or stream.
- A high DO level in a community water supply is good because it makes drinking water taste better. However, high DO levels speed up corrosion in water pipes. For this reason, industries use water with the least possible amount of dissolved oxygen.
- Adequate dissolved oxygen is necessary for good water quality. Oxygen is a necessary element to all forms of life. Natural stream purification processes require adequate oxygen levels in order to provide for aerobic life forms.
- The amount of DO affects what types of aquatic life are present in a stream, because many species of fish and macro invertebrates are sensitive to low DO levels. DO also regulate the availability of certain nutrients in the water.
- Most aquatic organisms require oxygen in specified concentration ranges for respiration and efficient metabolism, and DO concentration changes above or below this range can have adverse physiological effects.
- Short-lived anoxic & hypoxic events can cause major “kills” of aquatic organisms.
- Exposure to low oxygen concentrations can have an immune suppression effect on fish which can elevate their susceptibility to diseases for several years.
- the toxicity of many toxicants (lead, zinc, copper, cyanide, ammonia, hydrogen sulfide and pentachlorophenol) can double when DO is reduced from 10 to 5 mg L-1.
- If dissolved oxygen becomes depleted in bottom waters (or sediment), nitrification, and therefore de-nitrification, may be terminated, and bio-available orthophosphate and ammonium may be released from the sediment to the water column. These recycled nutrients can give rise to or reinforce algal blooms. Ammonia and hydrogen sulfide gas, also the result of anaerobic respiration, can be toxic to benthic organisms and fish assemblages in high concentrations.
- Total dissolved gas concentrations in water should not exceed 110 percent. Concentrations above this level can be harmful to aquatic life. Fish in waters containing excessive dissolved gases may suffer from “gas bubble disease”; however, this is a very rare occurrence.
4) What type of titration is involved in above test?
The Winkler method is also known as the iodometric technique because iodometric titration is used in its procedure and is considered the “gold standard” for measuring the concentration of dissolved oxygen in a sample of water. Through a series of chemical reactions, the O2 combines with iodine to form a golden yellow chemical. Therefore each oxygen molecule is associated with an iodine molecule, and we can measure oxygen by measuring the iodine. When the iodine is neutralized by the addition of sodium thiosulfate, the golden color disappears, and we can determine how much iodine (hence oxygen) was in the sample. (Note: some oxygen test kits use a starch indicator that turns the iodine solution from yellow to a deep blue color to make it easier to distinguish the color change.)
The chemistry of this test is based on the addition of a manganese solution followed up by a strong alkali solution. The DO present rapidly forms hydroxide salts with the manganese. The color of the floc formed is an initial indicator of how much DO is present. In the presence of iodide ions in an acidic solution, the oxidized manganese reverts back to the divalent state and the liberated iodine is equivalent to the original DO content. This iodine is then titrated with a standard thio-sulfate solution.
5) What is azide modification?
If nitrites NO2-1 will present in water, they will change the results because they convert iodide ions to iodine before performance of experiment. To remove NO2-1 sodium azides are added. Use the azide modification for most wastewater, effluent, and stream samples, especially if samples contain more than 50 mg NO2–-N/L and not more than 1 mg ferrous iron/L. The Azide Modification of the Winkler Method is the standard test for dissolved oxygen. In the analysis, manganous ion reacts with the dissolved oxygen present in the alkaline solution to form a manganese (IV) oxide hydroxide flocculent. Azide is then added to suppress interference from any nitrite, which would react with the iodide. The solution is then acidified and the manganese (IV) floc is reduced by iodide to produce free iodine as I3– in proportion to the oxygen concentration. The liberated iodine is then titrated to the starch-iodide end point. Dissolved Oxygen
Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen, Dissolved Oxygen
Thanks for sharing your thoughts on . Regards
I blog frequently and I genuinely thank you for your content.
Your article has truly peaked my interest. I’m going to take a note of your blog and keep checking for new information about
once a week. I subscribed to your RSS feed too.
I think the admin of this website is truly working hard in support of
his web site, for the reason that here every material is quality based stuff.
Thank YOU
As the admin of this site is working, no hesitation very rapidly it will be renowned,
due to its quality contents.
I hope
We are a bunch of volunteers and starting a new scheme in our community.
Your site provided us with valuable information to work
on. You have performed an impressive activity and our entire neighborhood will probably be
thankful to you.
thanks
I am trying to evaluate BOD of waste water and often I am seeing that before titration on adding starch the blue colour doesn’t develop at all. I am totally confused about this. Can anyone provide any probable reasons?
you can ask such questions here
https://www.facebook.com/groups/jobsforcivilengineers/?ref=group_header
thanks…………its really helpful
Welcome